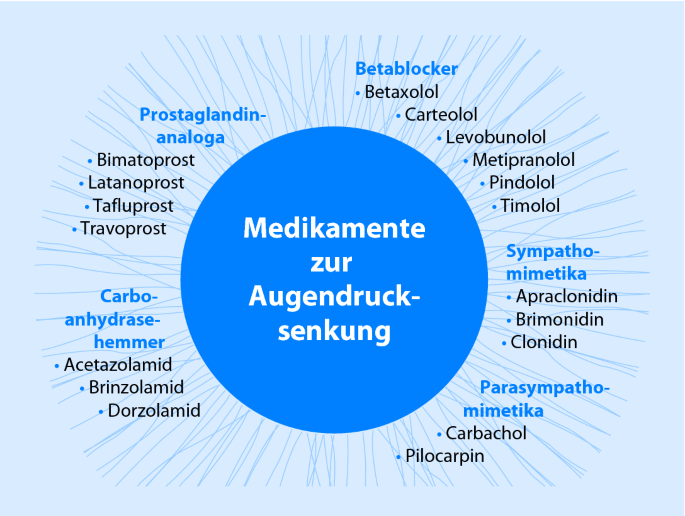
TAPTIQOM® Taflotan® sine Taflotan® COSDUO® Cosopt® sine Cosopt-S® Cosopt® Verschreibungspflichtige Arzneimittel

Dermatological adverse effects of anti‐glaucoma eye drops: a review - Patchinsky - 2022 - Journal of the European Academy of Dermatology and Venereology - Wiley Online Library
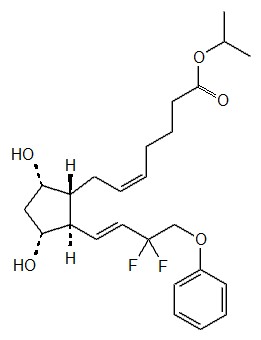
Decentralised Procedure Public Assessment Report Taptiqom Timolol maleate/ Tafluprost DE/H/3869/001/DC Applicant: Santen Oy Niit
Mutual Recognition Procedure Public Assessment Report Taflotan sine 15 Mikrogramm/ml Augentropfen Tafluprost DE/H/5250/001/MR Ap
Mutual Recognition Procedure Public Assessment Report Taflotan sine 15 Mikrogramm/ml Augentropfen Tafluprost DE/H/5250/001/MR Ap

Prevalence of ocular symptoms and signs with preserved and preservative free glaucoma medication | British Journal of Ophthalmology
TAPTIQOM® Taflotan® sine Taflotan® COSDUO® Cosopt® sine Cosopt-S® Cosopt® Verschreibungspflichtige Arzneimittel

DE102009021372A1 - Phosphate-free pharmaceutical composition for glaucoma treatment - Google Patents
TAPTIQOM® Taflotan® sine Taflotan® COSDUO® Cosopt® sine Cosopt-S® Cosopt® Verschreibungspflichtige Arzneimittel
Mutual Recognition Procedure Public Assessment Report Taflotan sine 15 Mikrogramm/ml Augentropfen Tafluprost DE/H/5250/001/MR Ap

DE102009021372A1 - Phosphate-free pharmaceutical composition for glaucoma treatment - Google Patents
Mutual Recognition Procedure Public Assessment Report Taflotan sine 15 Mikrogramm/ml Augentropfen Tafluprost DE/H/5250/001/MR Ap

PDF) Preservative-free bimatoprost 0.03% in patients with primary open-angle glaucoma or ocular hypertension in clinical practice